You've just finished teaching the Periodic Table to your chemistry or physical science students. You know you did an awesome job teaching periods, families, the periodic trends of electronegativity, atomic radius, and metallic character. You have no doubt that students know about the alkali metals, the halogens, and the noble gases. You are convinced that all the students will ace the unit test.
Then you notice a few glazed-over looks on the faces of the students. A review of the Periodic Table is needed!
Here's the Story:
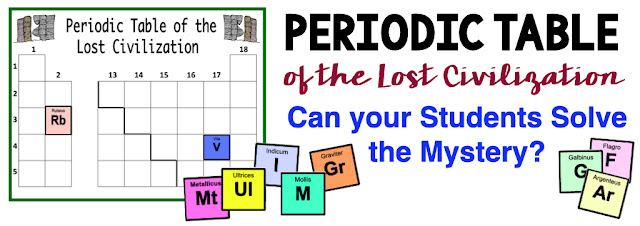
A long lost civilization has been found buried beneath many feet of lava from an ancient volcanic eruption. The population consisted of many scientists who were in the early stages of discovering and identifying the elements of the natural world. Unfortunately, a volcanic eruption destroyed the civilization before the periodic table was complete. The ancient civilization had different names and symbols for the elements than those used on the modern day periodic table. Students will read 28 statements that describe 34 elements from long ago. The statements give the element names, symbols, and information about the chemical and physical properties of the elements. Students must decipher the descriptions of the elements and place the elements in the correct position on the periodic table.
Some examples of the clues include:
- The following elements are known to be unreactive: Scansio (S), Lumen (Lu), Graviter (Gr), Piger (Pi) and Respiro (Rp). The element Scansio (S) has 2 valence electrons.
- Flavus (Fa) is the most reactive of the nonmetals.
- The atomic radius of Lumen (Lu) is much smaller than the atomic radius of Respiro (Rp).
- Piger (Pi) has valence electrons in the third energy level.
The students will work through the 28 clues and decide where to place the 34 elements on the periodic table of the lost civilization.
As stated above, the ultimate test to determine if students have mastered a concept is to see if they can apply their knowledge to a new situation. If students can arrange the 34 elements on the ancient period table, then they have a really good understanding of how the periodic table works.
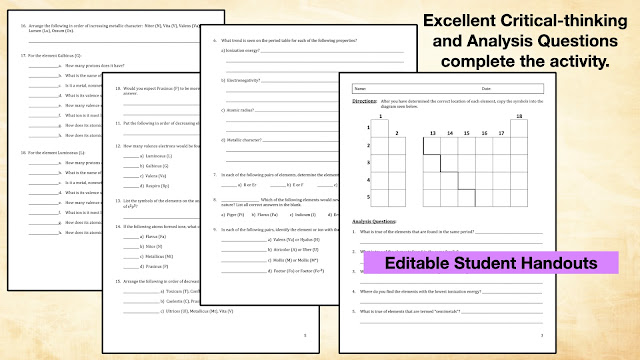
But, since this is a review activity, a 4-Page set of handouts is included with additional critical-thinking and analysis questions.
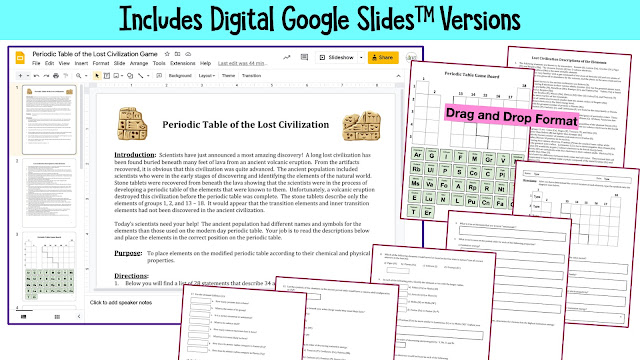
Putting pencil to paper is my preferred method of teaching, but if a digital version is needed, one is included. Students will "drag and drop" the element squares onto the ancient periodic table in Google Slides.
Students will always be more engaged and enthusiastic about reviewing for a test when the review is in a game format. This activity is highly engaging and fun for students. It's a blast to watch the light bulbs go on when students realize they have correctly identified the ancient elements. I hope your students will enjoy this activity as much as mine.
Have fun teaching!









No comments:
Post a Comment